



Urinary bladder catheterization is performed for both therapeutic, and perioperative purposes.
Catheter Associated Urinary Tract Infections [CAUTI] are one of the most common HAIs and about 75% of UTIs developed in hospitals are associated with a urinary catheter.
CAUTI are preventable by implementing infection prevention process and safe catheter insertion, maintenance, change and removal.
CDC recommends Antimicrobial Impregnated Foley Catheters for reducing CAUTI rates in healthcare[Category 1B].
Thanks to the scientists for their persistent research and innovation for the development of patented silver nanoparticles - NanoAgCide impregnated SilvoGuard Foley Balloon Catheter.
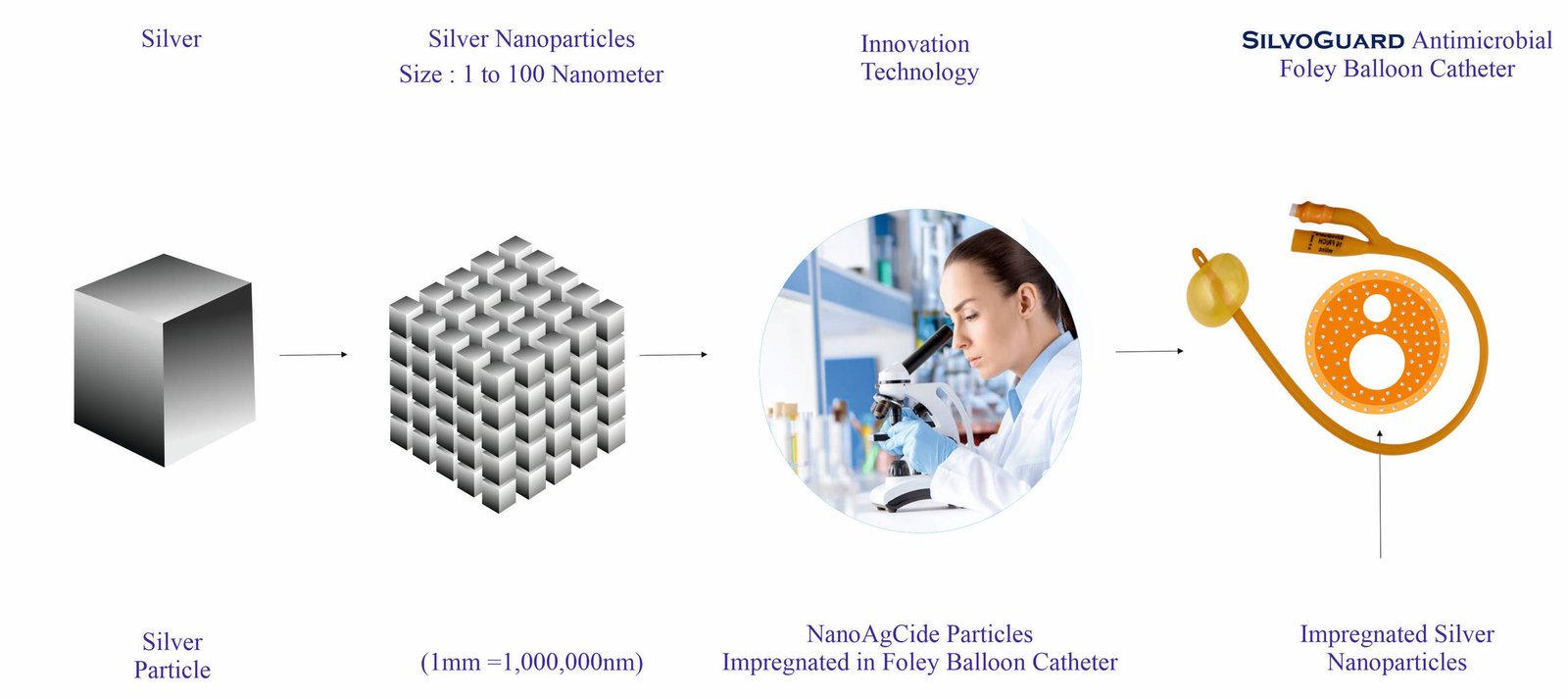
Government of India has awarded patent certification for the composition of silver nanoparticles and formulation there of. In the manufacturing process of SilvoGuard the NanoAgCide particles are impregnated in the urinary catheter material and are stabilized, uniformly compounded for their faster and better antimicrobial action.
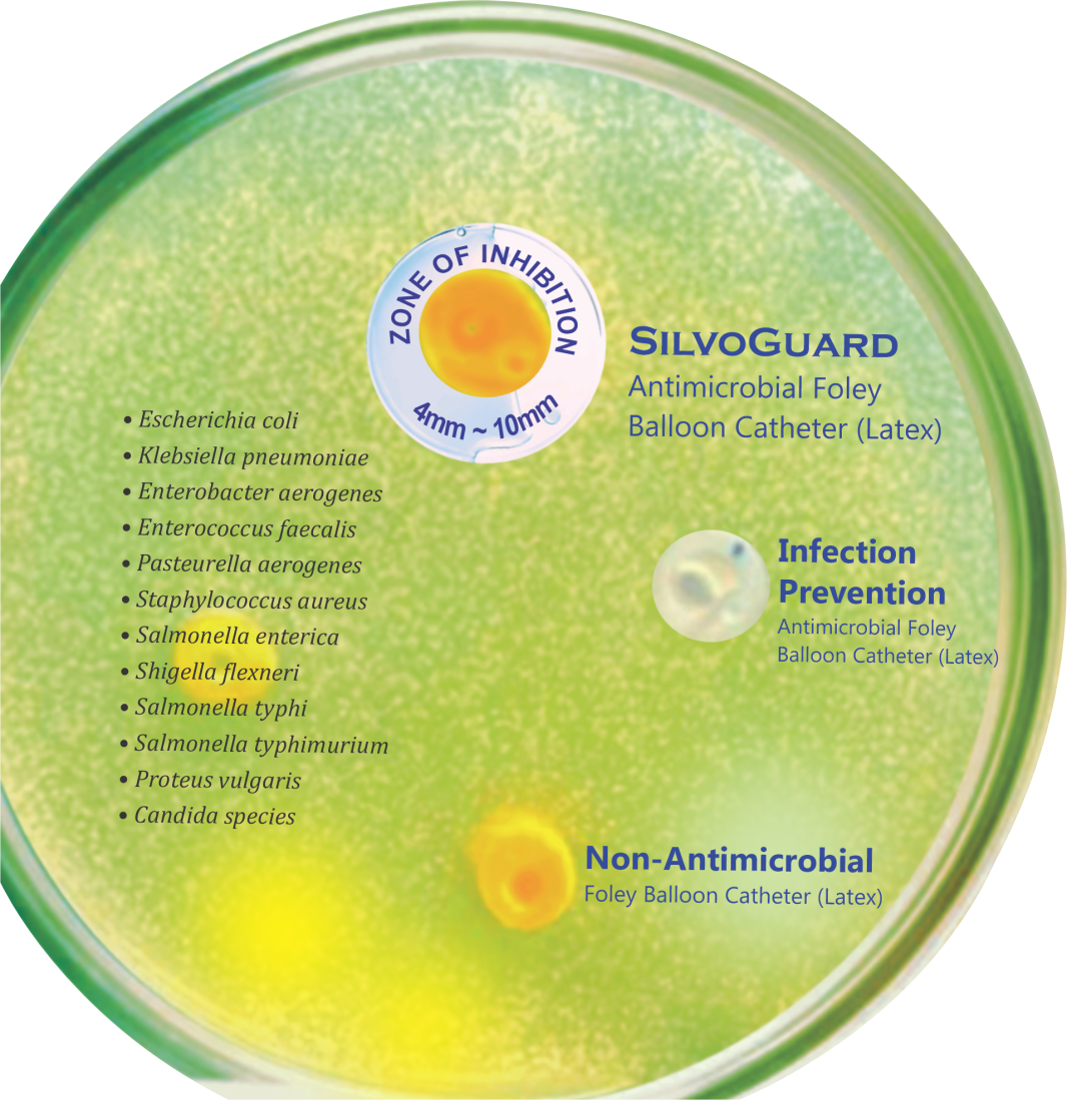
In vitro test, the reduction of microbial colonization by SilvoGuard Foley Balloon Catheter has been verified.
SilvoGuard exhibits superior antimicrobial action on the catheter surface.
It helps reduce CAUTI and improves patient care treatment.
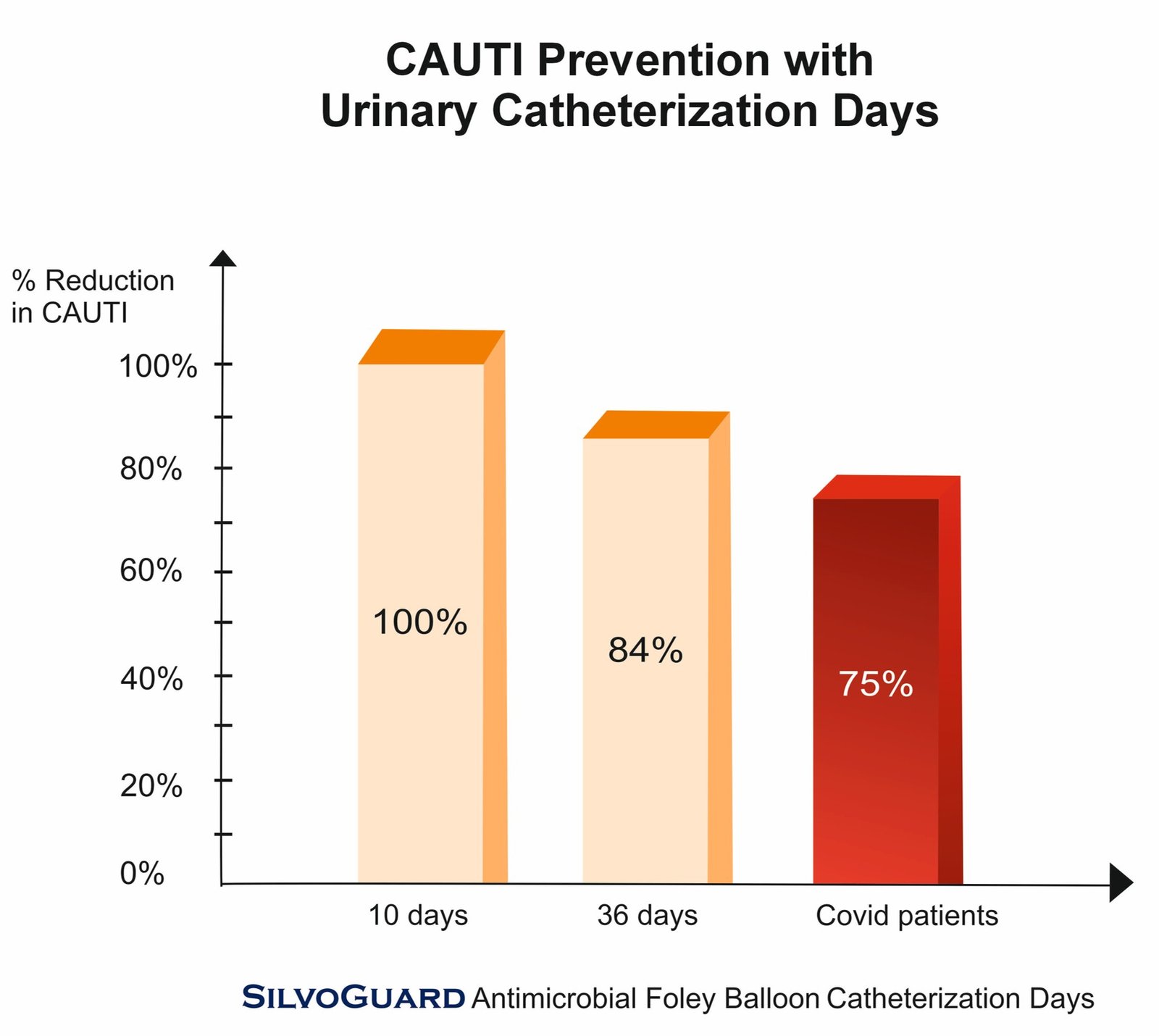
SilvoGuard Antimicrobial Foley Balloon Catheter is proven highly effective in the conducted studies for CAUTI prevention / reduction in the patients with short term and long term use of urinary catheterization days. Clinicians also experienced SilvoGuard to be non-toxic, easy to use and safe in nature.
Also, other problem related to use of catheters such as smell, biofilm formation, encrustation or catheter blockage were prevented.
SilvoGuard Antimicrobial Foley Balloon Catheter releases active silver ions for more than 40 days.



A  Group Company, specializes in the Manufacturing and Marketing of International-quality Medical Devices used for Anaesthesiology, Critical Care, Respiratory, Advanced Infection Management, Infusion Therapy and other Essential Medical Devices as well.
Group Company, specializes in the Manufacturing and Marketing of International-quality Medical Devices used for Anaesthesiology, Critical Care, Respiratory, Advanced Infection Management, Infusion Therapy and other Essential Medical Devices as well.

Registered Office :-
Unit No. 134, Damji Shamji Industrial Complex,
28, Mahal Industrial Estate, Mahakali Caves Road,
Andheri (East), Mumbai – 400 093, India.
Marketing Office :-
113 AB, Parwani House, Government Industrial Estate,
Charkop, Kandivali (West), Mumbai – 400 067, India.
Phone No.: +91 22-6830 7000
Customer Care + 91 8591663690
Email: info@lyvexmed.com